(It is an open access journal and everyone can download the whole contents. Click the above figure of the journal and move to the download site.)
The following is the summarized content. Sorry for a little difficulty in understanding due to specialty.
10 healthy aged individuals over 65 years old were enrolled. They were indicated to apply the lotion twice a day for 14 consecutive days on the neck. The skin was biopsied before and after the 14 days’ application and immmuno-histologically examined.
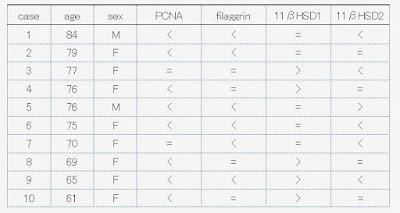
The results were as the table.
PCNA(proliferative cell nuclear antigen) is a marker of cell division and proliferation. Filaggrin is the main component protein of the corneal layer. 11βHSD1 and 2 are a little complicated to be explained. It has been revealed that the skin itself produces steroids (cortisol). The 11βHSD1 is associated with the activation of steroids while 11βHSD2 is with its inactivation. If 11βHSD1 decreases or 11βHSD2 increases after application of the hyaluronan lotion, it means that steroids within the epidermis is suppressed by the lotion and that can be one reason why the hyaluronan lotion make the atrophic skin recover.
The results show that PCNA increased in 8/10 cases, filaggrin increased in 6/10, 11βHSD1 decreased in 5/10 and 11βHSD2 increased in 5/10. The rate of the cases in which 11βHSD1 decreased or 11βHSD2 increased was 8/10.
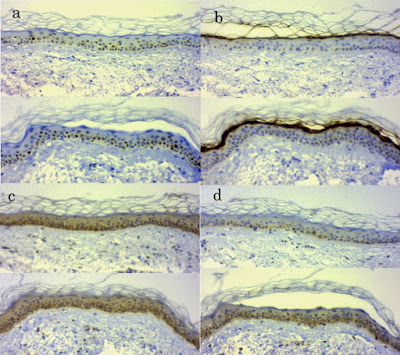
The real images of immunohistochemical staining are as follows in case 1 for example.
a: PCNA, b: filaggrin, c: 11βHSD1,d: 11βHSD2. Upper: before application, lower: after application. The brown color means positive findings.
From the above experiments, it was concluded that 1) Dr. Fukaya’s skin repair lotion has an efficacy of rejuvenating the aged skin. 2) The mechanism might be shifting 11βHSD1/2 balance.
Though the above study is about the aged skin of healthy individuals, it can be interpreted to the steroid-induced atrophy. It is because the atrophic skin due to steroids is also reported to recover by the same Dalton sized hyaluronan as the aged skin.
By the way, considering the fact that the aged skin and the atrophic skin due to steroids can both recover by the same hyaluronan, I suggest that the following can be said.
To apply topical steroids to the skin is very near to making the skin age.
The difference is that aging can’t be removed while topical steroids can be withdrawn. Anyway, I strogly recommend my hyaluronan lotion to all steroid-users. It might be a little expensive but it really works to prevent atrophy.
Don’t misunderstand me that I am only a merchant of my lotion. I am enough satisfied with my present successful situation and income as a cosmetic surgeon. The reason why I am transmitting information about TSA or suggesting my lotion is my outside interests.
Moreover, as a dermatologist also, it is really exciting to know the mechanism of the skin by which topical steroid addiction might be caused.
☆===☆=== ☆===☆===☆===☆
The folowings are addition in response to the comment of Javeriya Iftekhar.
Patients after long TSW period present more aged appearance than the real age because the swelling enlarges the skin especially around the eyelids.
Of course my lotion doesn’t improve this kind of sagging. The excessed skin should be excised surgically in such cases.
Just affter the upper eyelids surgery.

One week after the upper surgery and just after the lower eyelids surgery.
Two weeks after the upper surgery and one week after the lower eyelids surgery.
The price in my clinic is JPN 120,000 (about US$ 1,200) for the upper eyelids and JPN180,000 (about US$ 1,800) for the lower eyelids. I think they are absolutely reasonable. That is my everyday work. I don’t see patients with TSW or AD because I don’t like to charge money for only an advice. However, you are welcome if you like to make your appearance younger surgically.
This kind of work really keeps my mind healthy because I can charge money for my real skills and the result appears in only one week. It is too hard for doctors also to wait until the natural recovery comes with very few medical intervention. That is why I changed my work. But I am still transmitting information through internet like this because I know very well that my knowledge about TSW is precious for patients.
There is another option for improving finer lines. I will address it next time.
Please click here as for the clofibrate ointment. It is a non-steroidal agent with mild anti-inflammatory effect.
Sorry, the comment column is not available now. But the author believes readers can find some hints to overcome their own situations by the previous comments.